The chemical compound ammonium is formed in an acid-base reaction from the gaseous base ammonia by the addition of a proton to the free electron pair. Ammonia is a strong pungent smelling, colourless, water-soluble poisonous gas that irritates to tears and has a suffocating effect. Biologically, ammonia has an important function as an intermediate product in the breakdown of amino acids (basic building blocks for animal and plant proteins). In larger quantities, it is converted into the non-toxic urea for excretion in the body or into uric acid in birds. Ammonia is formed in larger quantities during the decomposition of dead living organisms or excrement by fungi and bacteria.
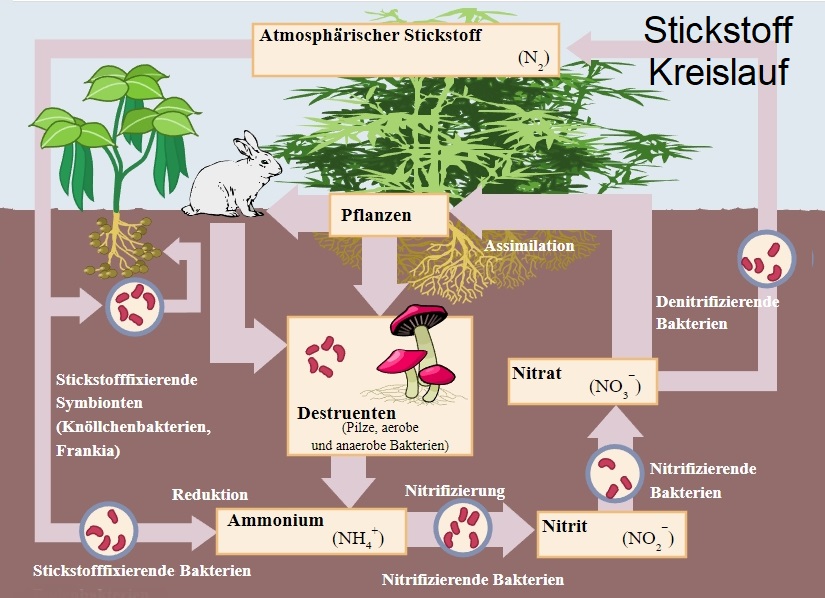
The natural formation of ammonia occurs from nitrogen in the air. This is a fundamental step in the course of the multi-stage process in the natural nitrogen cycle, which leads to the formation of nitrates with the help of nitrate bacteria. Artificial production takes place from the elements hydrogen and nitrogen. Most of the ammonia is processed into fertilisers, especially urea and ammonium salts. The two compounds ammonium sulphate (produced when ammonia reacts with sulphuric acid) and diammonium phosph ate (produced artificially from ammonia and phosphoric acid) are used as yeast nutrient salts to support fermentation. A deficiency of ammonium in the soil can lead to various vine diseases such as stem rot.
Graphic: By User:Nojhan, edited original Nitrogen, CC BY-SA 3.0, link.
Voices of our members

There is a vast number of sources on the web where one can acquire knowledge about wine. But none has the scope, timeliness and accuracy of the information in the encyclopaedia at wein.plus. I use it regularly and rely on it.
Sigi Hiss
freier Autor und Weinberater (Fine, Vinum u.a.), Bad Krozingen