Element (P) that is luminous through oxidation and whose name is derived from phosphoros (Greek for light bearer). In nature, it occurs exclusively bound, mostly in the form of phosphates. Typical minerals are apatite, phosphorite, turquoise and wavellite. It is also contained in guano (bird excrement) and is used in this form as fertiliser. Pure, white phosphorus produces corrosive sores and is very toxic; as little as 50 mg is fatal to humans. Phosphorus compounds are essential for all living organisms and are involved in central areas such as DNA as a component of nucleic acids and cellular energy supply. In the metabolism of the vine, phosphorus fulfils a variety of functions. It plays a central role in the energy balance, in photosynthesis and as a component of proteins and enzymes. The proportion is particularly high in the grape seeds. In very simplified terms, it can be said that phosphorus primarily serves the formation of blossoms and fruit.

In strongly acidic and also alkaline soils, phosphorus can become fixed (bound) and is thus no longer available to plants. A phosphorus deficiency in the vine is manifested by a blue-green to purple discolouration of the leaves and weak flower and fruit formation. Since the end of the 19th century, large amounts of phosphorus fertiliser have been added to most soils used for viticulture. A deficient supply of phosphorus to the vine is therefore not fundamentally due to a deficiency in the soil, but often to low availability. This is because the phosphates tend to form compounds that are difficult to dissolve and thus difficult to make available. Only a small proportion of the phosphorus in the soil is present in water-soluble, absorbable form. Fertilisation with phosphates must therefore be precisely adapted to the soil depending on the pH value. Phosphates are also used as insecticides for pest control.
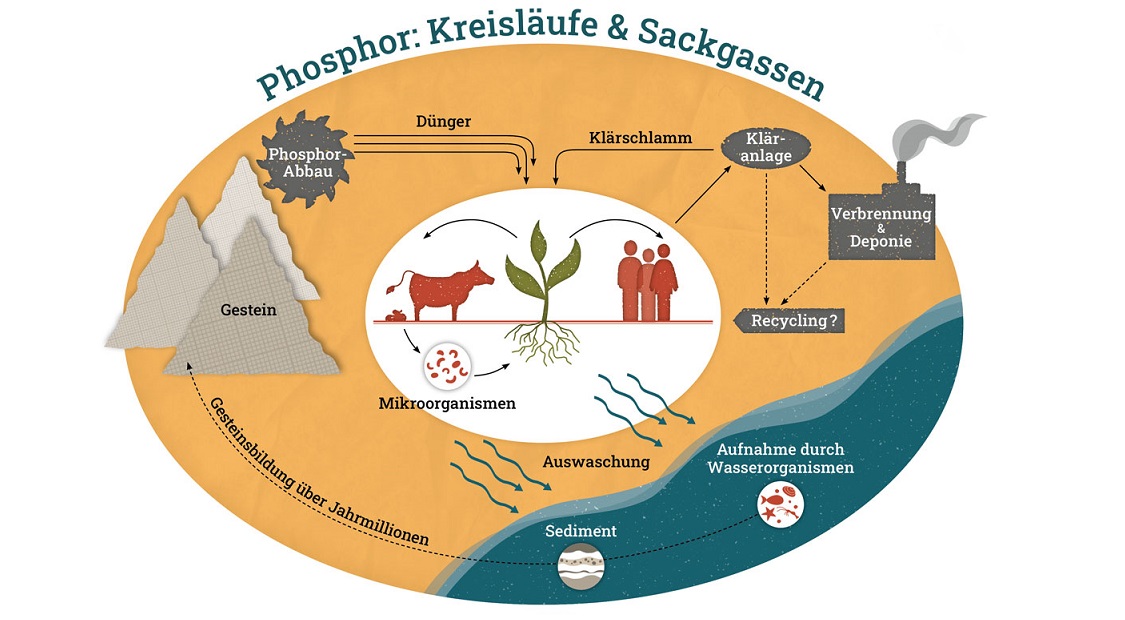
Graphic: by HGMedien/K.Peick - Own work, CC BY-SA 4.0, Link
Phosphorus: from CC BY-SA 3.0, Link
Voices of our members

The glossary is a monumental achievement and one of the most important contributions to wine knowledge. Of all the encyclopaedias I use on the subject of wine, it is by far the most important. That was the case ten years ago and it hasn't changed since.
Andreas Essl
Autor, Modena