The most important acid (also 2,3-dihydroxysuccinic acid or 2,3-dihydroxybutanedioic acid) in wine with a content of 0.5 to 4 g/l, which is one of the non-volatile acids. Before the discovery of the actual tartaric acid, its salt potassium hydrogen tartrate - the tartar - was thought to be a solid acid, since this precipitates easily in wine due to its poor solubility in water. In the past, this was therefore easier for chemists to recognise than the highly soluble tartaric acid. This was separated from tartar by the chemist Karl Wilhelm Scheele (1742-1786) in 1769 and was therefore named tartaric acid by him. After its continuous build-up in the grapes during the ripening period, the proportion no longer decreases, in contrast to malic acid. During prolonged cold weather, tartaric acid can already be precipitated in the grape.
Tartaric acid is by far the most sour-tasting acid in wine. It is approved in the EU as food additive E 334. It is also used to acidify low-acid wines in order to increase the acidity, subject to the country-specific, wine-law regulations. Metatartaric acid is esterified tartaric acid that is added shortly before bottling to prevent the precipitation of tartar or to stabilise it. The total content of total acid in wine is indicated with the collective name tartaric acid, although other acids are also contained. A derivative of tartaric acid is grape acid. See a list of all wine constituents under total extract.
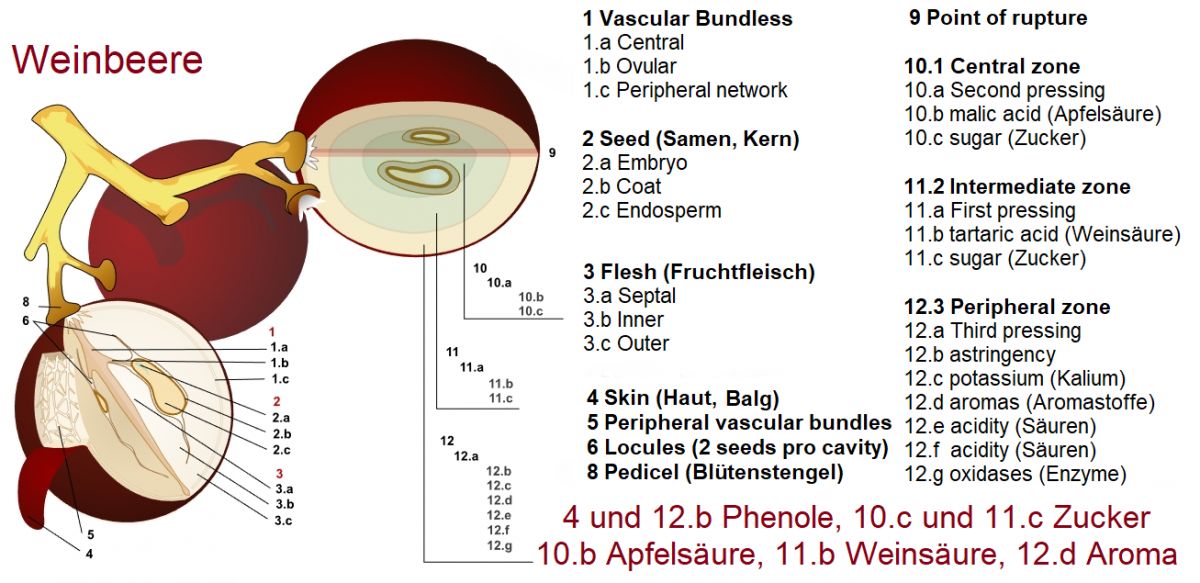
Graphic: By Mariana Ruiz Villarreal(LadyofHats), public domain, link
edited by Norbert Tischelmayer, March 2019
Voices of our members

The Wine lexicon helps me to keep up to date and refresh my knowledge. Thank you for this Lexicon that will never end in terms of topicality! That's what makes it so exciting to come back often.
Thorsten Rahn
Restaurantleiter, Sommelier, Weindozent und Autor; Dresden